Interventional Care


We notice that you are visiting us from . This site only services US-based visitors. Would you like to visit the site that is appropriate for your location?

At PDI, we provide innovative products and solutions, educational resources, in-service training, and clinical support to help you in the fight against preventable infections.
Advancing our products with a commitment to environmental and social responsibility, prioritizing safety, quality, and innovation to protect and enhance lives.
Learn how PDI’s Profend swabs can protect surgical and ICU patients from infections, the impact it made at Hunterdon Medical Center in New Jersey.
Tru-D® SmartUVC, a PDI Healthcare solution, has launched the next generation of intelligent UVC disinfection products. The Tru-D iQ system adapts to the needs of a wide range of room sizes to administer a precise dose of UVC.
Designed to clean external areas on energized electronic equipment and compatible with equipment surfaces that require a 70% IPA formulation.

At PDI, we provide innovative products and solutions, educational resources, in-service training, and clinical support to help you in the fight against preventable infections.
Learn More!Healthcare associated infections (HAIs) can increase the length of the hospital stay, require readmission or transfer to a higher level of care. Even worse, HAIs can lead to sepsis or death.

Clean and disinfect surfaces in every environment.
See Product Family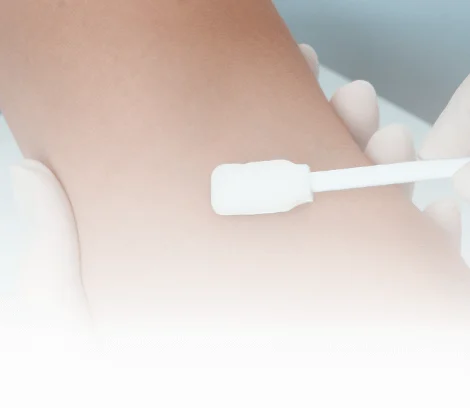
Provide skin antisepsis, device disinfection and nasal decolonization.
See Product Family
Keep patients clean & comfortable with personal hygiene & cleansing products.
See Product Family
Tru-D delivers UVC light to inactivate microorganisms on nonporous surfaces.
See Product FamilyHealthcare facilities need multiple layers of defense, combining products, guidelines, and techniques—because no single approach can fully eliminate the risk of HAIs.*
*statement is not reflective of the Tru-D device.










Beyond the Sneeze: The Role of Surface Transmission with Respiratory Viruses


Earn free CE Credits while learning from industry experts.

Discover white papers, videos, case studies, & more.
Search our database of over 1,200 pieces of equipment.
Keep up-to-date on the latest trends from industry experts.

Keep up-to-date on the latest trends from industry experts.
This course qualifies for 1 CE / 1 IPU and corresponds to the following Infection Prevention Unit (IPU) domain(s): Management…
This course qualifies for 1 CE / 1 IPU and corresponds to the following Infection Prevention Unit (IPU) domain(s): Identification…
This course qualifies for 1 CE / 1 IPU and corresponds to the following Infection Prevention Unit (IPU) domain(s): Identification…
This course qualifies for 1 CE / 1 IPU and corresponds to the following Infection Prevention Unit (IPU) domain(s): Identification…
Given the rapidly increasing number of procedures performed in this setting, national programs to measure and improve the quality of care provided in ASCs are essential.

Behavioral healthcare represents an often overlooked and underrepresented area in healthcare. To understand the challenges of infection prevention in this setting, PDI Clinical Science Liaison, Amanda Thornton, RN, MSN, CIC, VA-BC, sits down with Emma Parsons, BSN, RN, CTSS, owner and Director of Nursing of Sequoia Behavioral Health (3) to tackle the topic.
Prior to 2020, there had been a significant ongoing and sustained reduction in the rate of central line-associated bloodstream infections…

What can the Infection Preventionist do to help mitigate the infection risks within LTC? Here are a few important points to remember when approaching the environment of care.
Our 40-year heritage has exclusively focused on fighting preventable infections and preserving the well-being of the facilities, clinicians and patients touched by our products.
Infection prevention is our passion—it’s all we do.
LEARN MORE






References: